Alzheimer's, Aduhelm, and the amyloid hypothesis.
Why the new Alzheimer's drug was approved by fast track by the US FDA when it shows no cognitive benefit for patients and the scientific history behind it.
Alzheimer’s disease (AD) is one of the biggest public health crises we are facing today. It’s the 7th leading cause of death in the US and the number of patients suffering is growing rapidly. As we get better at treating heart disease, diabetes, and cancer, more and more people will live with and die of Alzheimer’s. The risk of becoming diagnosed with AD grows exponentially after about age 70, and if you live to be 95 years old, you have a 50/50 chance of having it. Patients typically suffer for 3-13 years depending on age at diagnosis, spend $360,000 on care, and >80% of the care giving falls on friends and families to shoulder. So understandably, everyone really wants a drug that works for AD.
Until 2021, and the most recent approval of Biogen’s Aduhelm, the only drugs approved for AD were four Acetylcholinesterase (AChE) inhibitors (tacrine, rivastigmine, galantamine, and donepezil) and the NMDA receptor antagonist, memantine. ACh and NMDA are neurotransmitters and AChE inhibitors and memantine increase and decrease neuronal signaling respectively. They attempt to compensate for or protect against the AD-related death of specific neuron populations in the brain. Unfortunately, neither class of drug does much to improve symptoms. The combination of memantine and donepezil has been shown to be "of statistically significant but clinically marginal effectiveness". Imagine someone who has a cognitive score of 7/30 (severe AD) on a memory test designed for the average person to easily pass, and memantine raises that score to 7.5 or 8/30. That small bump in memory for someone with that severe of AD won’t do much to help them remember anything about what they did today, or to remember who their daughter or son-in-law is, or to keep them safe from wandering out of the house. Some people would argue that any treatment is better than no treatment. However, there are side effects in many users— upset stomach and vomiting being the most common, and less frequently, muscle cramps, decreased heart rate, decreased appetite and weight, hallucinations, confusion, dizziness, headache, and fatigue. These side effects in combination with these drugs’ marginal efficacy makes it questionable whether they are worth taking for some patients.
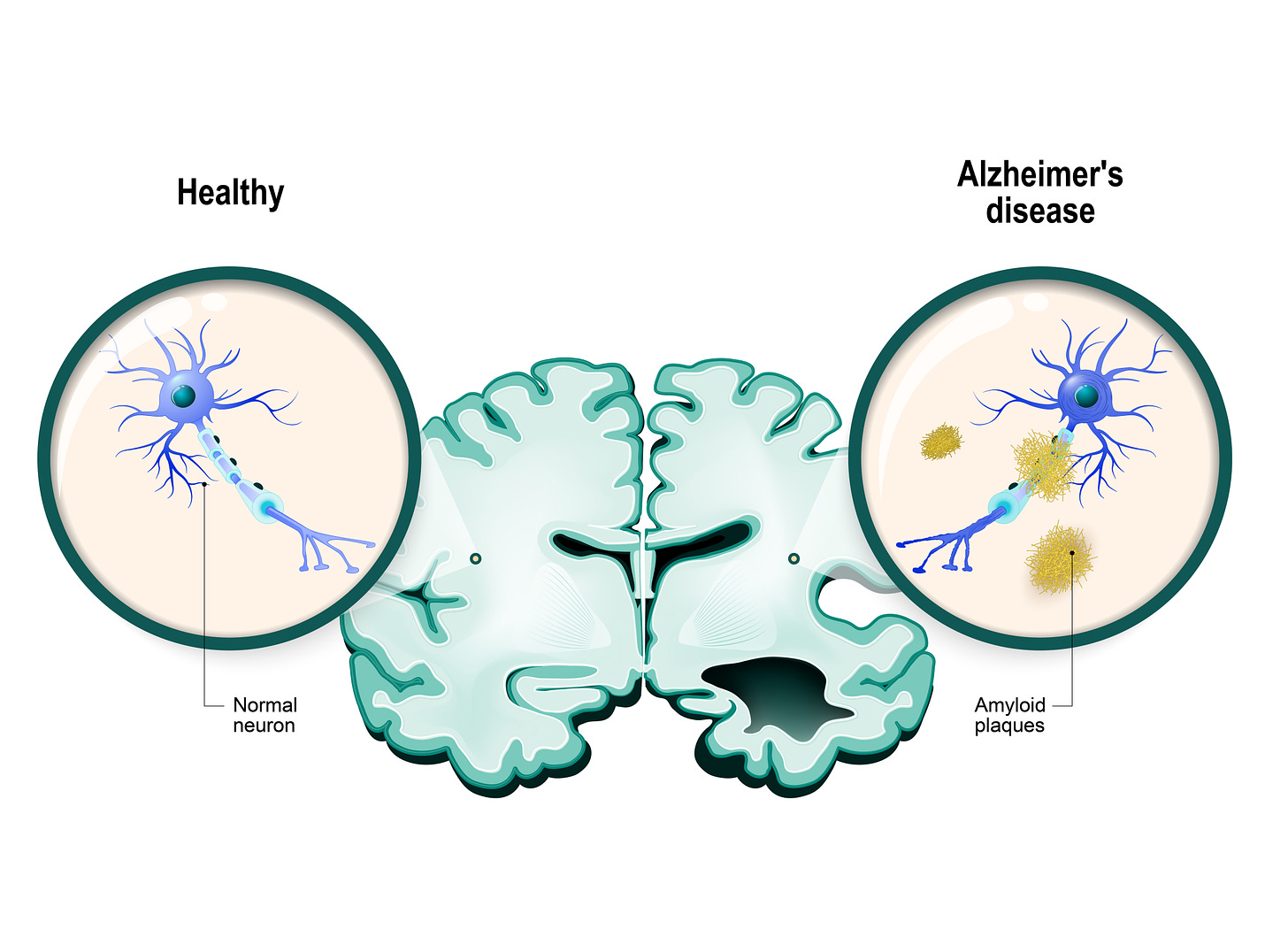
Many of the attempts at creating new drugs, including Biogen’s controversial, Aduhelm, have been centered around “plaque busting”. Plaques are misfolded proteins that are deposited between neurons in AD. AD is primarily caused by neurons dying in a particular area of the brain, the hippocampus. Plaques are also found in this regional pattern in people with AD. This area of the brain is responsible for shorter term memory formation and is the reason that AD patients first lose short term memory and then progress to losing longer term memory, which is stored in many regions of the brain and thus remains intact until the very latest stages of AD. Patients will remember their childhoods but not their breakfast for instance, for this reason.
All neurodegenerative diseases basically boil down to neurons dying in one region of the CNS or another. In Parkinson’s dopaminergic neurons die. In ALS, specific neurons in the spinal cord die. This regional patterning of cell death is particularly interesting as it points to something that is cell type specific to each disorder, but I digress.
Scientists have long hypothesized that plaques, which are made up of the protein amyloid, may be causing AD— causing neurons to die in the hippocampus. A lot of excitement was generated from a 1999 paper in Nature by Elan Pharmaceuticals showing that AD mice could be treated with anti-amyloid immunotherapy to restore normal memory. The problem with this paper and with drug validation in mice for neurodegenerative diseases in general is that mice aren’t humans and they don’t normally get AD or plaques in their brains. Causing mice to have memory problems by injecting them with human disease genes is a far from perfect way to determine what might happen in humans as this could be causing problems in mouse cognition that have very little to do with AD.
The amyloid hypothesis also fits with a top AD genetic risk factor, APOe4, which increases the probability of getting AD by 8-12x— as APOe4 is involved with the processing of amyloid. However, there are many people who have a lot of plaques in their brains, which we can see by special imaging of the brain for plaques (PET-CT), who are completely cognitively normal. If plaques were the sole cause of AD then this could not be the case. This has led many scientists, including myself, to believe that the amyloid hypothesis is a red herring— that plaques are somewhat correlated with having AD but are not the cause of it. However, it’s possible that plaques are one of several causes of AD and protective factors can compensate for plaques in people that have them but are cognitively normal. This is the key debate with plaque busters like Aduhelm. Aduhelm is an IV antibody therapy which clears plaques from the brain. It does a very good job of this when looking at CSF and brain imaging.
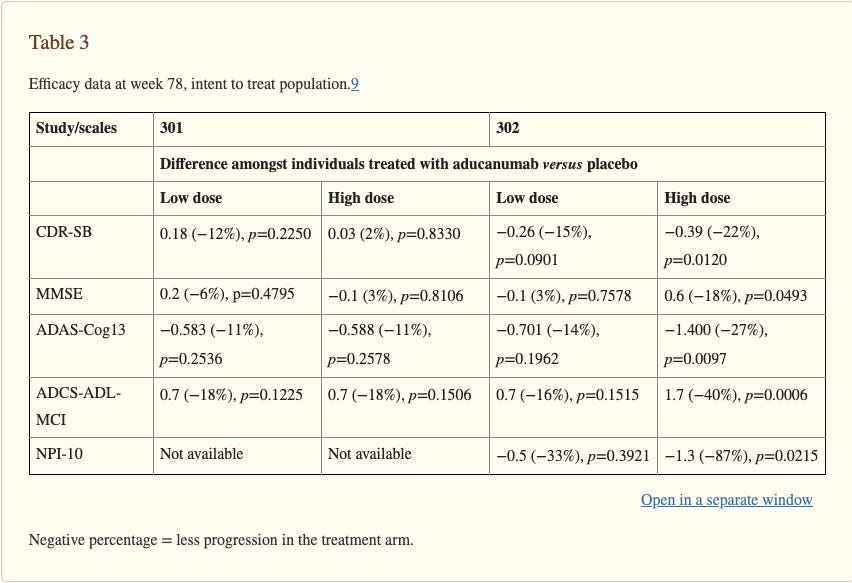
However, here’s the kicker, in clinical trials (~3,200 patients with mild cognitive impairment (MCI) or mild AD, MMSE 24-30) there was no statistical improvement in cognition after removing the plaques in one of the trials and marginal benefit at the highest dose in the second trial. The relevant variable to look at in the below is MMSE (Mini Mental State Exam)— a 30 point test of memory given to patients to test for AD.
There have also been prior attempts by other companies at creating plaque busters, and so far none of them have worked.
Aduhelm was approved in July 2021 (excerpt from FDA press release below:
" accelerated approval pathway which can be used for a drug for a serious or life-threatening illness that provides a meaningful therapeutic advantage over existing treatments. Accelerated approval can be based on the drug’s effect on a surrogate endpoint that is reasonably likely to predict a clinical benefit to patients, with a required post-approval trial to verify that the drug provides the expected clinical benefit."
This was based on efficacy for biomarkers— ie. plaques being reduced. I question why plaques were allowable as biomarkers instead of the clinical correlate that is actually relevant, rate of cognitive decline. In part this may be a definitional problem— neurologists define AD as the presence of brain pathology including plaques— however since we don’t fully understand AD, this could be a biologically irrelevant way to define it. This is a problem that many psychiatric disorder definitions also suffer from.
The FDA went against a special committee-the Peripheral and Central Nervous System (PCNS) Drugs Advisory Committee- that voted 10 (against)-1 (undecided) not to approve Aduhelm and three members of that committee resigned over the decision. The thinking was to give Biogen another 9 years to provide more evidence that the drug improves cognition or slows disease progression. Patient advocate groups are additionally strongly lobbying for Aduhelm’s approval. It has, however, been pointed out that many of these groups are funded by biotech companies that are creating antibody therapies, so there may be some conflict of interest in these lobbying efforts.
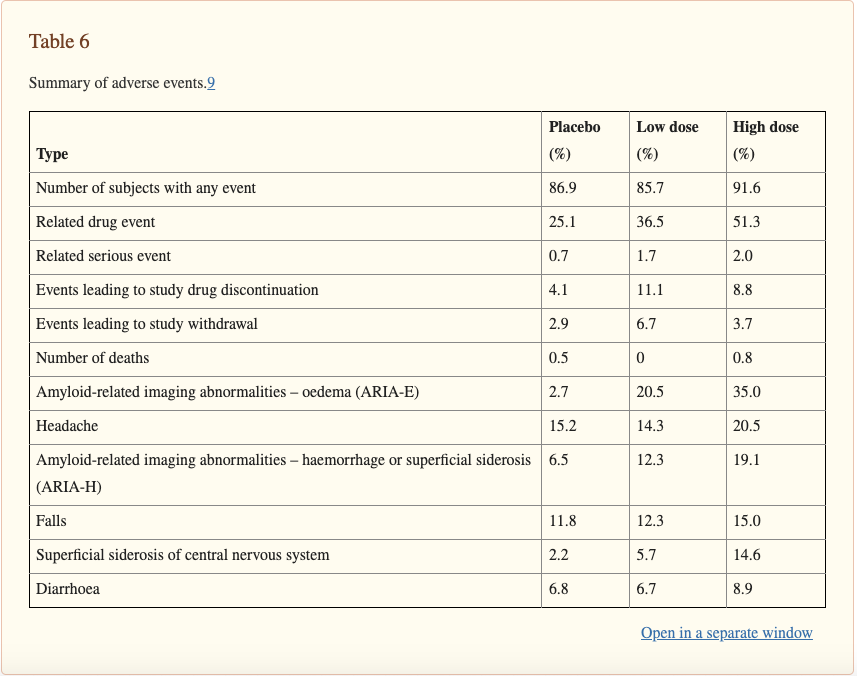
There are a few potential harms to giving patients Aduhelm to consider, for one there are some serious side effects noted in the clinical trial including ARIA (swelling of the brain) and brain bleeds.
Administering IV drugs is also potentially dangerous for infection risk and uncomfortable for patients who may already be agitated and not understand the treatment that they are receiving. Additionally, the exclusion criteria for patients in the trial is extensive enough that many physicians may not be comfortable prescribing the drug because their patients have conditions that excluded them from the trial including stroke, mental health conditions, and cardiovascular conditions.
Aduhelm also originally came with a price tag of ~$56,000/year, which recently has been lowered to about half of that by Biogen. If prescribed widely, this would ~1.5x existing medicare spending for a potentially dangerous drug with no proven cognitive benefit. So far, physicians and hospital systems have not been quick to administer Aduhelm and drug sales are way below expectation.
I personally think it was a mistake to approve Aduhelm because I am concerned that in 9 years when the drug fails to provide any benefit to patients because it was designed on a faulty premise, there will be a large backlash that could set us back in creating AD therapies that actually do work. It could make investors wary of backing companies, such as mine, that are working to create efficacious therapies that prevent neuron death and improve cognition. One thing is clear though, approval of Aduhelm should finally put the debate over the amyloid hypothesis to rest one way or another.
Stay tuned for future posts to learn the best ways that we have currently have to keep your memory sharp and prevent AD— and what future therapies that work for AD may look like. Hint- you might want to pour yourself a cup of coffee, break out that treadmill, and start working on the other biggest risk factor for AD besides APOe4, aging.
If you want to support this work, please consider subscribing to this newsletter and get the added benefit of chatting with me in the comments. You can also find me on Twitter @DrGlorioso.