How to Decrease Your Late Stage Cancer Risk from 20% to 2%
In 2003, Steve Jobs was diagnosed at age 48 with a pancreatic neuroendocrine tumor, a rare and slower-growing form of pancreatic cancer. His tumor was found by accident on a CT scan for kidney stones. At diagnosis it had not spread, and this type of tumor caught early is often curable with surgery. But Jobs delayed the recommended surgery for nine months in favor of alternative therapies, and by the time he consented, the cancer had reached his liver. He died in 2011 at age 56. No screening guideline would have flagged his cancer. Annual full body MRI likely would have.
In late 2022, Susan Wojcicki, the former CEO of YouTube, was diagnosed with non-small cell lung cancer (the most common type) at age 54. It had already spread. She had never smoked, was running several miles a day, and had almost no symptoms. Under current US guidelines, she would never have qualified for a lung CT scan, because every guideline restricts lung screening to people with significant smoking histories. She died in August 2024 at age 56.
In 2013, Beau Biden was diagnosed with glioblastoma at age 43, the most aggressive type of brain cancer. He had no symptoms until a sudden episode of disorientation during a family vacation. He underwent surgery, radiation, and chemotherapy, but the cancer came back. He died in 2015 at age 46. There is no recommended screening for brain cancer and no blood test that detects it, but many glioblastomas evolve from slower-growing brain tumors that grow silently for years, and annual brain MRI can catch those during the window when intervention matters most.
All three died in their forties or fifties from cancers that screening guidelines were not designed to find. The uncomfortable reality about cancer screening in 2026 is that even the most proactive patients have enormous blind spots, and most people don’t realize how much of their cancer risk is completely unmonitored.
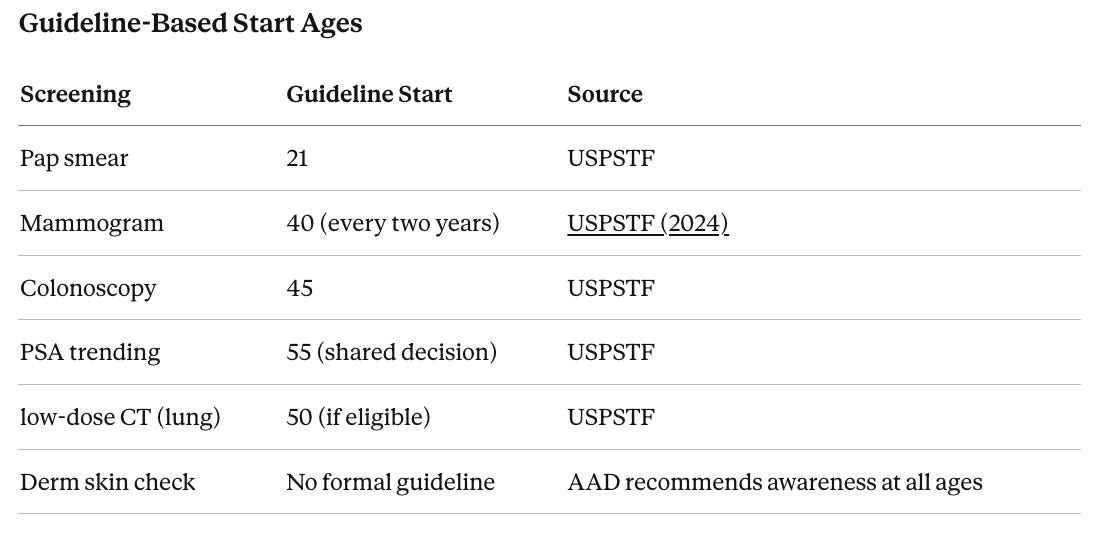
What Standard Screening Actually Covers
The average American has access to five evidence-based cancer screenings, each targeting a single cancer type.
Mammography screens for breast cancer, the most common cancer in women with a roughly 13% lifetime risk. The US Preventive Services Task Force (USPSTF) updated its recommendation in 2024 to begin screening every two years at age 40, and many doctors recommend annual screening. Mammography catches roughly 85-90% of breast cancers per screen. For women with BRCA1 or BRCA2 mutations (inherited DNA changes that dramatically increase breast and ovarian cancer risk), guidelines recommend annual breast MRI beginning at age 25, because BRCA carriers face a 40-72% lifetime breast cancer risk and their tumors tend to develop younger.
Pap smear (often done alongside an HPV test, which checks for the virus that causes most cervical cancers) screens for cervical cancer (~0.6% lifetime risk). With regular screening, cervical cancer is almost entirely preventable because the test catches abnormal cell changes years before they turn into cancer.
Colonoscopy screens for colorectal cancer (~4% lifetime risk). Guidelines recommend starting at age 45, with repeat every 10 years if no polyps are found. Colonoscopy is unique because it both detects and prevents cancer by removing precancerous polyps (small growths on the colon lining that can turn cancerous), reducing colorectal cancer risk by roughly 60-70%.
PSA testing (prostate-specific antigen, a protein made by the prostate gland that can be measured in blood) screens for prostate cancer (~12.5% lifetime risk in men). The USPSTF categorizes PSA as a shared decision-making conversation rather than a routine recommendation, citing concerns about overdiagnosis (finding cancers that would never have caused harm). The ERSPC trial (a large European study) demonstrated that PSA screening saves lives, and overdiagnosis estimates of 20-50% are largely a product of how the test was historically used, with biopsies (procedures where a tissue sample is removed and examined for cancer) triggered automatically whenever PSA rose above 4.0. Tracking PSA over time, where the speed of rise is monitored across years rather than a single number prompting immediate action, substantially improves the ability to distinguish aggressive cancers from slow-growing ones. A baseline PSA at 35 followed by repeat measurements creates a risk picture that a single test at 55 cannot replicate.
Low-dose CT screens for lung cancer (~6% lifetime risk). The National Lung Screening Trial demonstrated a 15-20% reduction in lung cancer deaths with annual low-dose CT (a type of X-ray that uses very little radiation) in high-risk adults (ages 55-80 with a 20+ pack-year smoking history, meaning one pack per day for 20 years or equivalent). US guidelines currently restrict low-dose CT to significant smokers, leaving the vast majority unscreened for the leading cause of cancer death.
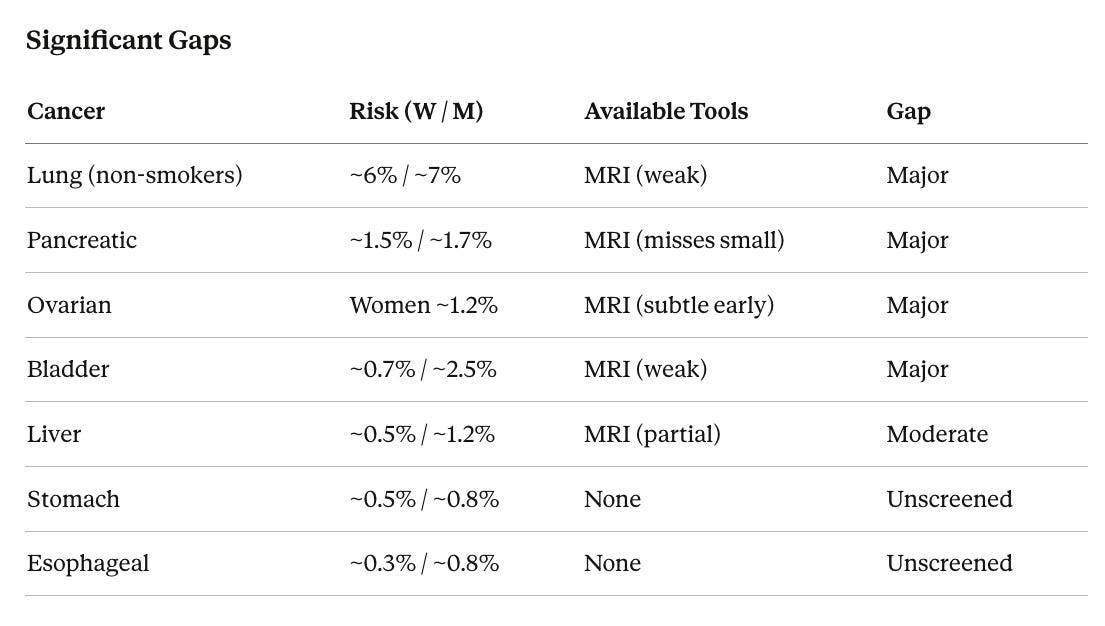
That is where guideline-based screening ends. Five cancer types. Everything else, including pancreatic, ovarian, brain, liver, stomach, esophageal, kidney, bladder, and dozens of others, has no recommended screening whatsoever.
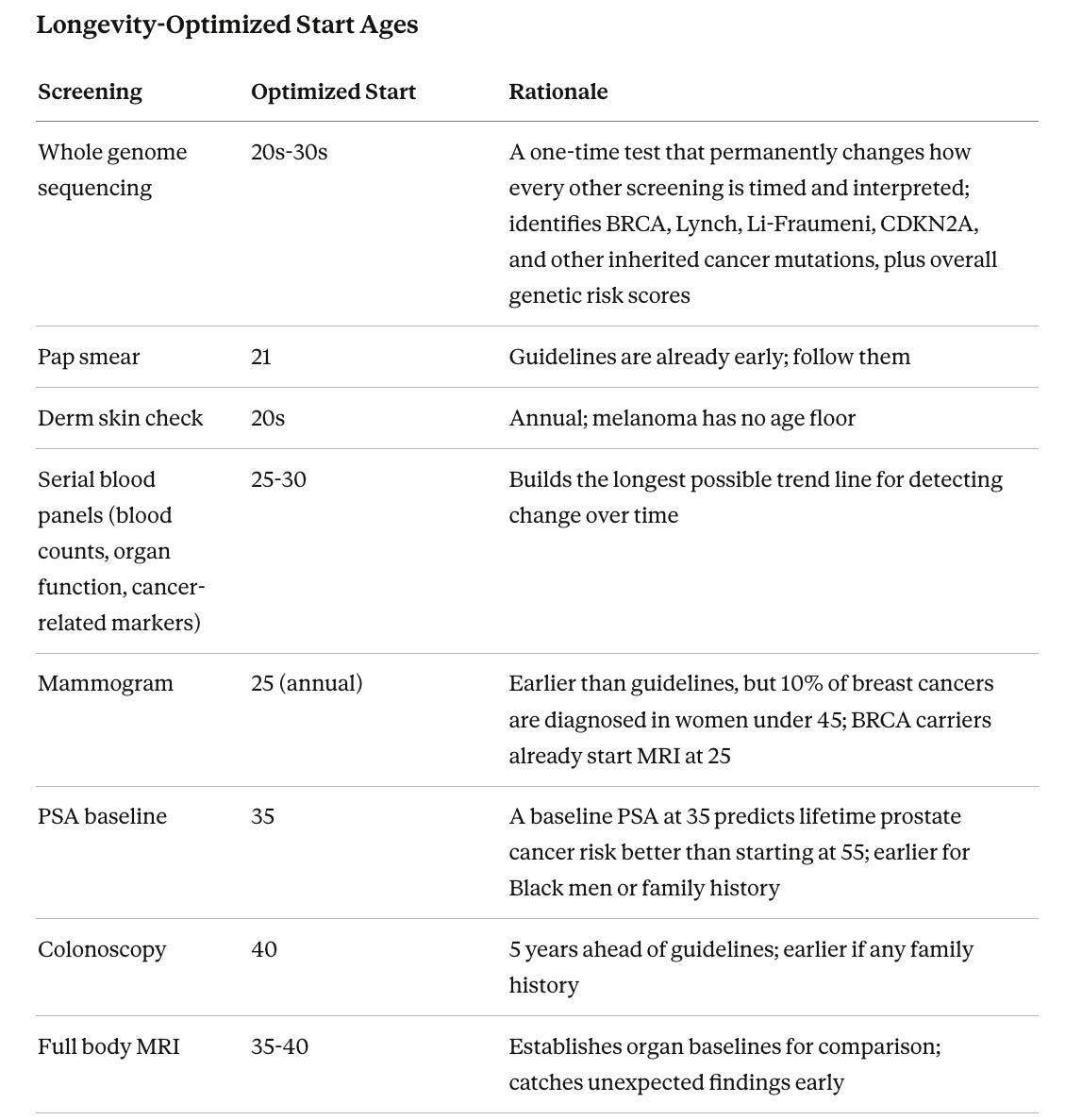
When to Start
Guidelines represent minimum recommendations for average-risk populations. For anyone pursuing longevity, the question is not “when do guidelines say I should start?” but “when does starting earlier meaningfully change my odds?”
These guidelines weigh false positives, overdiagnosis, and healthcare costs against lives saved. They are deliberately conservative and based on averages that do not account for the 34-year-old diagnosed with breast cancer or the 38-year-old with Stage III colorectal cancer. Roughly 10% of breast cancers occur in women under 45, and colorectal cancer in adults under 50 has been rising for two decades.
The logic is straightforward. For tools that track trends over time (blood panels, PSA), every year of baseline data makes changes easier to spot. For imaging (mammogram, MRI), earlier starts mean earlier baselines to compare against. For colonoscopy, starting earlier extends the window for removing precancerous polyps before they become dangerous. The trade-off is more false positives, more follow-up tests, more anxiety, and more cost. But for individuals who understand that trade-off, earlier screening is a rational choice no guideline explicitly prohibits.
For people with strong family histories, particularly pancreatic, ovarian, colorectal, or breast cancer diagnosed before 50 in a parent or sibling, screening should begin even earlier. Genetic testing (discussed below) can clarify the right timeline.
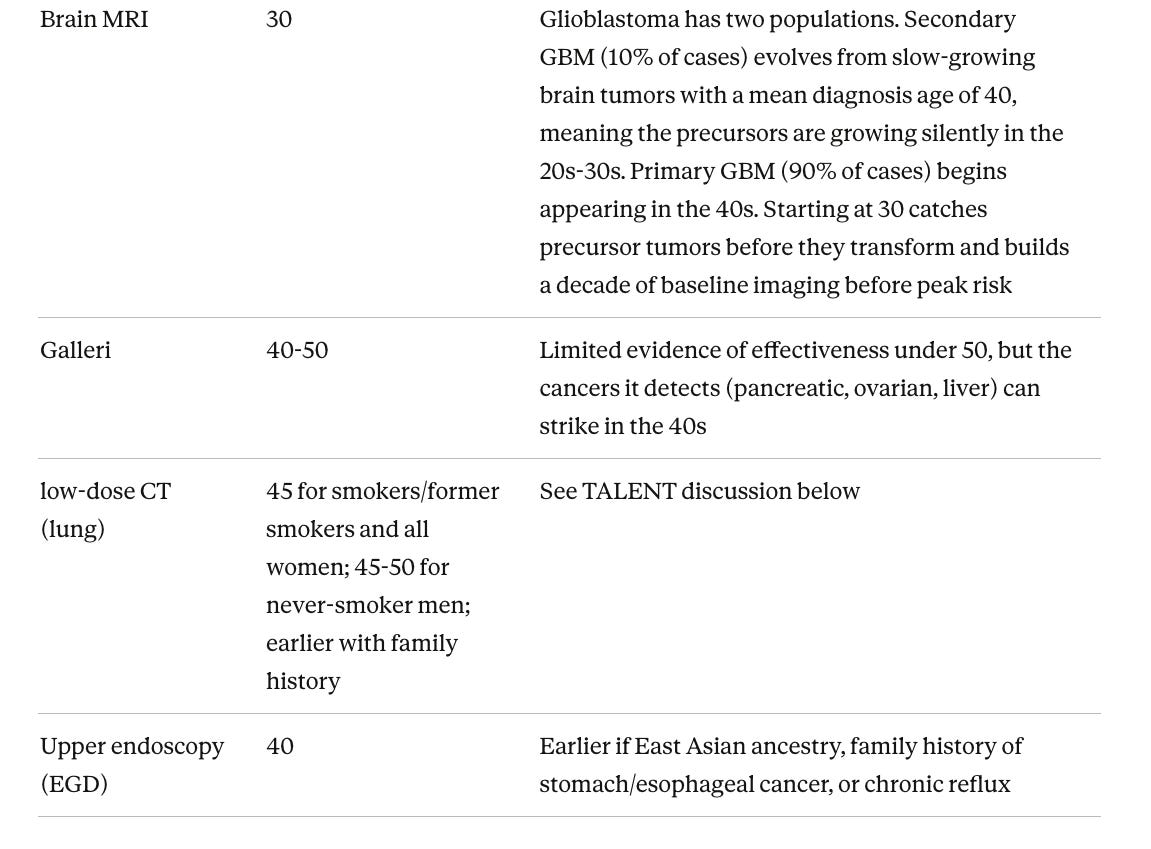
A note on low-dose CT and never-smokers. Every US guideline restricts lung CT to smokers and former smokers because the landmark trials (the NLST and Dutch-Belgian NELSON trial) only enrolled heavy smokers. The TALENT trial from Taiwan changed this picture by screening 12,011 never-smokers aged 55-75 with risk factors. The cancer detection rate was 2.6%, more than double the NLST (1.1%), and 96.5% of cancers found were stage 0 or I (meaning potentially curable). The strongest predictor was family history, with risk scaling from 2.0% with no family history to 9.1% with four or more affected relatives. Being female and being over 60 each increased risk independently. Taiwan became the first country to offer subsidized lung CT screening to never-smokers with family history.
There are important caveats. Lung cancers in never-smokers are biologically different, mostly a subtype called adenocarcinoma (a cancer starting in the cells lining the inside of organs), and some may never become life-threatening. The false positive rate was high (17.4%), and long-term data on whether this screening actually reduces deaths is not yet available. Still, for never-smokers with family history, the data supports low-dose CT every two years starting around 45. Even participants without family history had a 2.0% detection rate, nearly double the NLST. Given that lung cancer is the leading cause of cancer death and never-smoker lung cancer is rising, particularly in women, low-dose CT every two years starting at 45 is a reasonable consideration for anyone optimizing for longevity.
What a Full Body MRI Adds
A growing number of people are adding annual full body MRI through services like Prenuvo. MRI can detect solid organ masses in the liver, kidneys, pancreas, ovaries, uterus, and brain, along with some lymphomas and bone growths. For liver and kidney tumors, MRI is excellent. For ovarian and uterine masses, it provides meaningful detection.
Brain cancer is where MRI is uniquely valuable. Glioblastoma, the most common cancerous brain tumor, has a five-year survival rate of just 4.7%. There is no blood test and no other imaging that reliably finds it. But many glioblastomas evolve from slower-growing tumors that grow silently for years, and MRI can catch those precursors during the window when treatment has the greatest impact. Services like NeuroAge pair brain MRI with cognitive testing and genetic risk assessment for integrated brain health monitoring.
The limitation is that MRI is not equally good at all cancer types. Small pancreatic tumors are frequently missed, early ovarian cancer is subtle, lung nodules are far better detected by CT, and cancers lining hollow organs (stomach, esophagus, bladder) are largely invisible on MRI. Still, as Jobs’s case shows, even imperfect detection matters when there is no other screening option.
MRI does not catch melanoma (you need a dermatologist) or blood cancers like leukemia (no mass to image). Thyroid cancer can show up on full body MRI if the protocol includes neck coverage, though ultrasound remains the dedicated tool for thyroid evaluation.
What Blood Panels Add
Serial blood testing through services like Function Health adds another layer, though its direct cancer screening value is modest. Individual markers like CA-125 (linked to ovarian cancer), CEA (linked to colorectal and pancreatic cancer), and AFP (linked to liver cancer) produce too many false alarms as standalone screens. But tracking them over time catches meaningful changes that a single snapshot would miss.
Blood panels are genuinely useful for catching blood cancers. A complete blood count (CBC, which measures all the different types of cells in your blood) can flag leukemia through abnormal white blood cell counts, unexplained anemia (low red blood cells), or low platelets (the cells that help blood clot). Chronic leukemias are often first identified as surprise findings on routine bloodwork showing a gradually rising white cell count. Multiple myeloma (a cancer of plasma cells, immune cells that make antibodies) can be flagged by a blood test called serum protein electrophoresis. Lymphoma (cancer of the lymph nodes), however, often has a completely normal blood count and is better caught by imaging.
How Genetics Changes the Screening Equation
Every screening tool discussed so far operates on the assumption that the person has average population risk. Genetic testing upends that assumption. It is the only tool in the screening stack that permanently changes how every other tool is used.
The highest-impact findings are inherited DNA changes (passed from parent to child) that dramatically increase lifetime risk for specific cancers. BRCA1 and BRCA2 carriers face a 40-72% lifetime risk of breast cancer and 11-44% risk of ovarian cancer. Lynch syndrome (mutations in DNA repair genes MLH1, MSH2, MSH6, or PMS2) carries up to 78% lifetime risk of colorectal cancer, 60% risk of uterine cancer, and elevated risk of ovarian, stomach, and brain cancers. Li-Fraumeni syndrome (TP53 gene mutations) carries roughly 90% lifetime cancer risk across multiple organs. CDKN2A mutations substantially increase melanoma and pancreatic cancer risk. Each of these triggers dramatically earlier and more intensive screening.
These are not rare edge cases. A 2024 study of nearly 27,000 adults found that 1.3% carried one of these high-risk mutations, and 90% did not know it before testing. A Lynch syndrome carrier who starts colonoscopy at 45 has already been accumulating unmonitored colorectal cancer risk for two decades.
Beyond high-risk mutations, polygenic risk scores add up the effects of hundreds of common genetic variations into a single number estimating where someone falls on the risk spectrum. Think of it like height: no single gene makes you tall, but hundreds together determine your height. Cancer risk works similarly. For prostate cancer, the BARCODE1 trial in the New England Journal of Medicine showed that screening men in the top 10% of genetic risk detected aggressive cancers that PSA and MRI alone would have missed.
Genetic testing also enables prevention, not just better screening. BRCA1/2 carriers who have both breasts removed preventively reduce breast cancer risk by 90-95%, and adding removal of ovaries and fallopian tubes reduces ovarian cancer risk by roughly 80%. Lynch carriers who have a preventive hysterectomy (removal of the uterus) essentially eliminate uterine cancer risk, and enhanced colonoscopy every 1-2 years starting at 20-25 reduces colorectal cancer deaths by roughly 65-72%. For a BRCA1 carrier, these interventions can take a 40-72% lifetime breast cancer risk down to the low single digits. Cancer-preventing medications like tamoxifen or aromatase inhibitors (drugs that block hormones that fuel certain breast cancers) can further reduce risk.
For anyone building a screening stack, genetic testing should be one of the first steps. Whole genome sequencing captures every known high-risk cancer mutation, enables polygenic risk score calculation, and never needs repeating because the raw data can be reanalyzed as science advances. Services like NeuroAge pair genetic risk assessment with brain MRI and cognitive testing for genetically informed brain health monitoring.
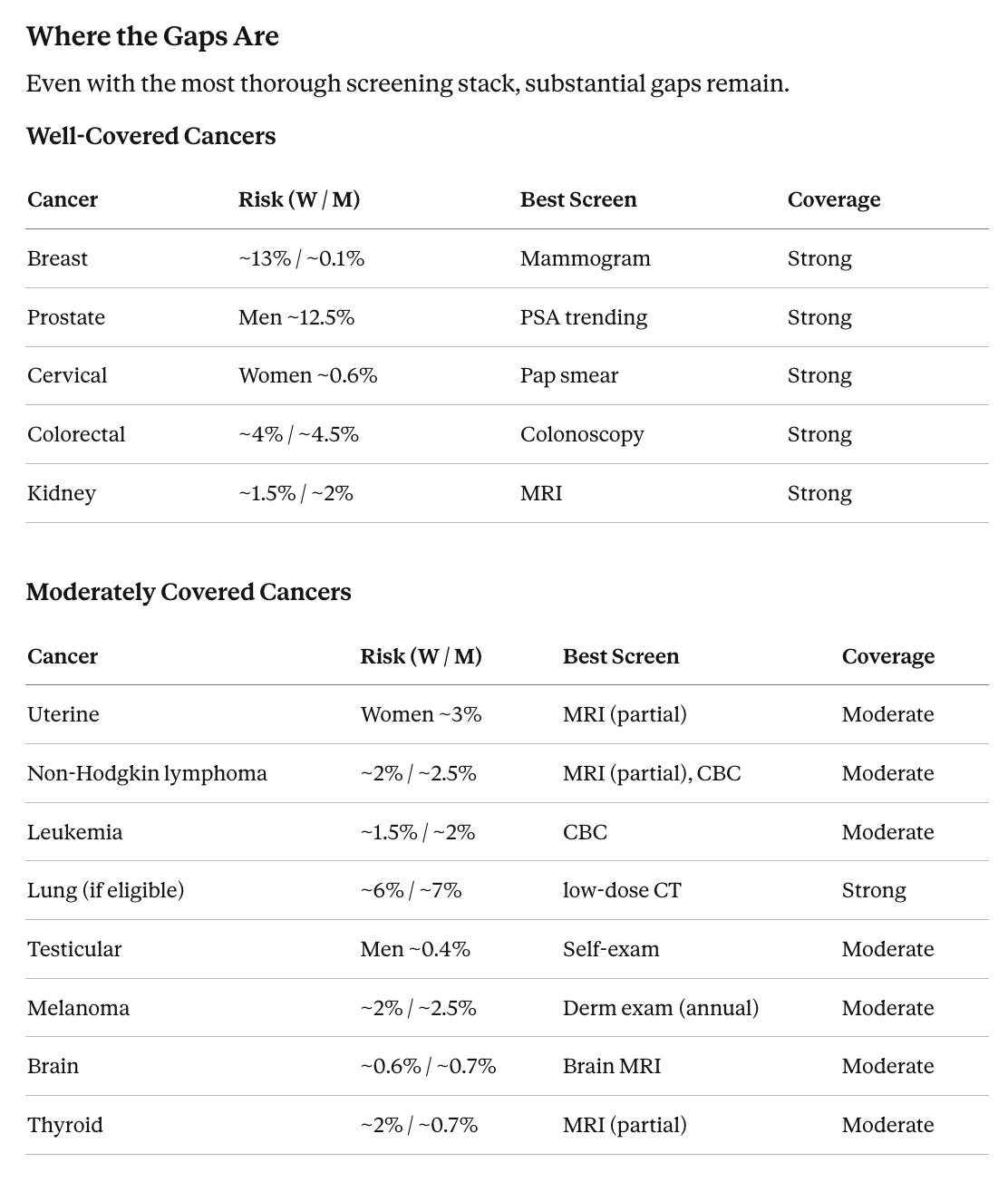
Even with every tool listed above, roughly half your total cancer risk is either unscreened or poorly screened. The biggest gaps are lung cancer in non-smokers, bladder, stomach, and esophageal cancer, along with imperfect coverage for pancreatic and ovarian cancer.
What Galleri Adds to the Stack
Galleri screens for more than 50 cancer types from a single blood draw. It works by analyzing chemical tags on tiny fragments of DNA that tumors shed into the bloodstream. Different cancer types leave different patterns, which allows Galleri to both detect a cancer signal and predict where in the body it is likely located. For in depth analysis of Galleri testing, read my Substack article, “The New York Times Called Galleri a Failed Test. I Think They Got It Wrong.”
For cancers with no existing screening (stomach, esophageal, bladder, and several blood cancers), Galleri (and similar tests in development) are the only blood-based detection option. For pancreatic and ovarian cancer, it adds a completely independent detection method alongside MRI. In the PATHFINDER 2 study (35,878 participants, presented at ESMO 2025, a major international cancer research conference), Galleri correctly identified 73.7% of the 12 deadliest cancers, with a false alarm rate of just 0.4%. Three-quarters of the cancers detected had no recommended screening test. For Stage I cancers specifically, Galleri caught approximately 61.9% of pancreatic, 50.0% of ovarian, and 100% of liver/bile duct cancers, based on Grail’s data from their earlier accuracy study.
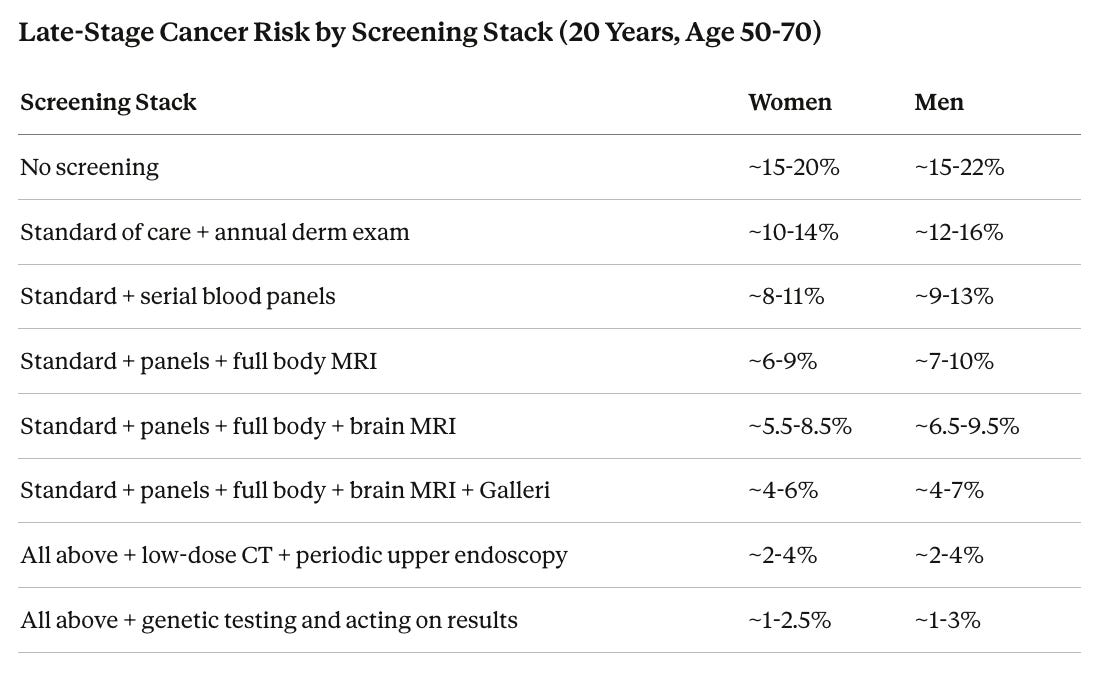
How the Math Changes at Each Layer
For a 50-year-old planning the next 20 years, here is a rough estimate of the probability of a cancer reaching Stage III or IV before detection.
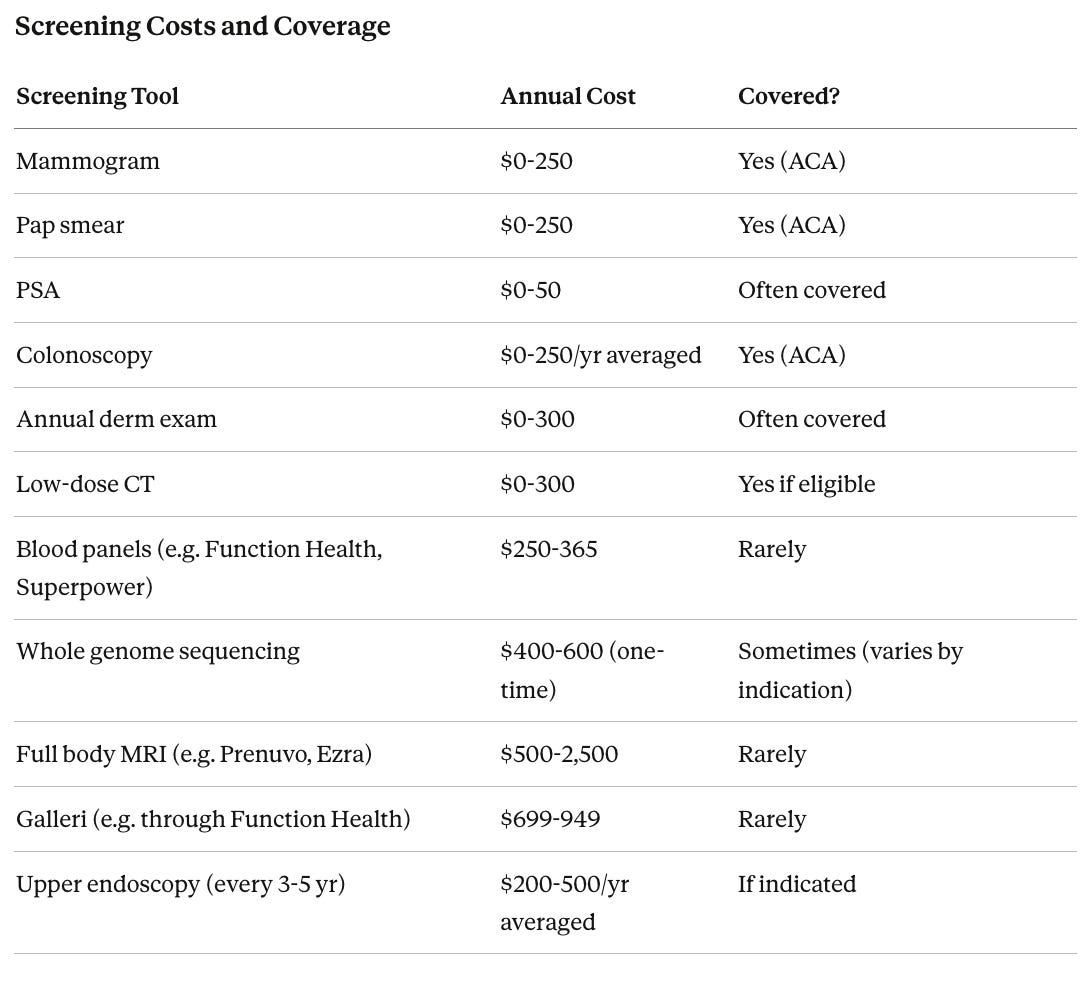
“Standard of care” for women includes mammogram (annual, covered under the Affordable Care Act), Pap smear with HPV testing (every 3-5 years, ACA-covered), colonoscopy (every 5-10 years starting at 45, ACA-covered), and annual skin check (typically covered). For men it includes PSA tracking (typically covered ages 55-69), colonoscopy, and skin check. For significant smokers, annual low-dose CT is strongly recommended (covered per USPSTF) and has been proven to save lives per the NLST and NELSON trials. The add-on layers (blood panels, MRI, Galleri) are rarely covered and run roughly $1,450-3,800/year. Upper endoscopy (EGD, where a thin camera is passed down the throat to examine the esophagus and stomach) is typically covered when a doctor orders it. The final row reflects genetic testing followed by acting on results. For the ~1.3% carrying high-risk inherited cancer mutations, this can include preventive surgery, increased screening frequency, and cancer-preventing medications.
These are estimates based on published detection data, cancer stage data from SEER (the federal government’s main cancer statistics database), and the assumption that each screening layer catches some cancers others miss. Individual risk varies based on genetics, family history, and lifestyle.
One often-overlooked addition is upper endoscopy every 3-5 years, which is the only way to directly examine the esophagus and stomach lining. It is standard screening in Japan and South Korea and worth discussing with a gastroenterologist (a doctor specializing in the digestive system).
The Risks and Costs of Screening
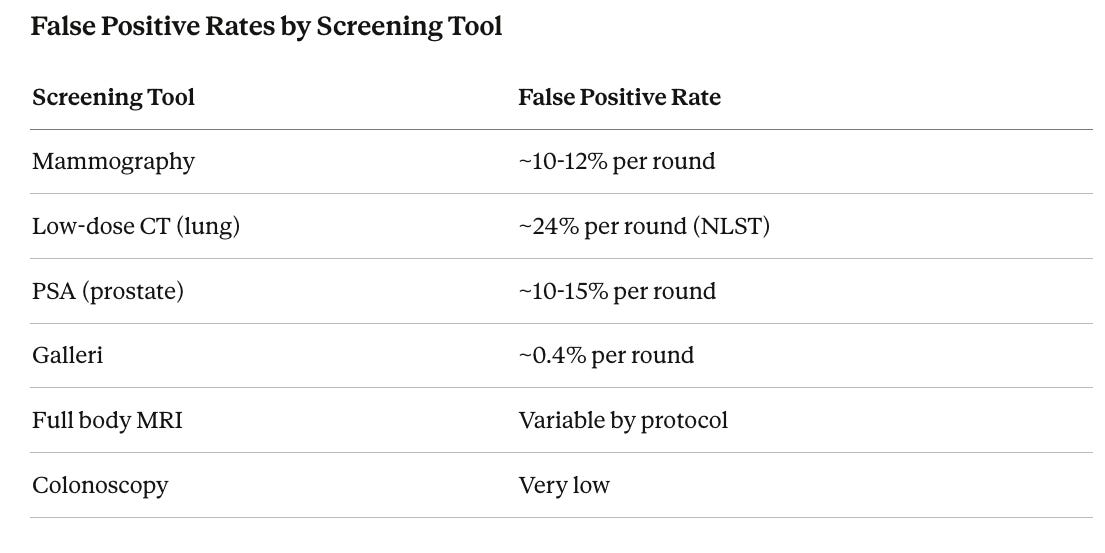
Radiation exposure is a concern primarily with lung low-dose CT. A single scan delivers roughly 1.5 millisieverts (mSv) of radiation. For comparison, the average person absorbs about 3 mSv per year from natural background sources. Twenty years of annual screening adds roughly 30 mSv total, estimated to cause approximately 1 additional cancer per 2,500 people screened. MRI, Galleri, and mammography (0.4 mSv) involve minimal to no radiation.
False positives are the most common harm. Each positive triggers follow-up tests, specialist referrals, biopsies, anxiety, and cost.
Overdiagnosis (detecting cancers that would never cause symptoms or death) is most relevant to prostate cancer (estimated 20-50% with PSA), thyroid cancer, and some slow-growing breast cancers. Although if you are a longevity enthusiast or someone with supercentenarian genetics, you might live a decade (or three) longer than the average person so these tumors that you would normally “die with” will become real issues at older ages. The commonly cited “overdiagnosis concern” only makes sense if you die at 80, not 120.
Galleri appears to preferentially detect aggressive cancers, which may reduce overdiagnosis, though this needs more data.
For out-of-pocket items (MRI, Galleri, blood panels), the annual cost is roughly $1,450-3,800, or $29,000-76,000 over 20 years. As a comparison point, the annual probability of a house fire is roughly 0.25%, and most people carry homeowner’s insurance without question against a risk smaller than the 3-4% chance of screening catching a cancer that would otherwise kill you.
How Much Cancer Is Preventable
Not all cancer risk requires screening to manage. According to the American Cancer Society, approximately 40% of cancer cases and 45% of cancer deaths are linked to controllable risk factors, led by cigarette smoking (19%), excess body weight (8%), alcohol (5%), UV radiation (5%), physical inactivity (3%), and cancer-causing infections like HPV and hepatitis B and C.
For someone who has never smoked, maintains a healthy weight, exercises regularly, limits alcohol, uses sun protection, and is vaccinated against HPV and hepatitis B, the 35% lifetime cancer risk drops to roughly 20-25%. The remaining cancers tend to be driven by genetics, random DNA copying errors that accumulate with age, and aging itself. Prevention and screening are complementary. A clean lifestyle reduces the probability you develop cancer. Screening reduces the probability a cancer you do develop kills you.
The Hard Floor
With every screening tool available in 2026, the maximum stack combined with optimized lifestyle brings the probability of a late-stage cancer diagnosis down to roughly 1.5-3% over 20 years. It does not reach zero, for three reasons. First, interval cancers. Some aggressive tumors develop between annual screening rounds. Second, no test catches everything, and rare cancers can slip through multiple tools simultaneously. Third, some cancers are simply hard to find early, including cancers lining the digestive tract, early bladder cancer, some blood cancers, and glioblastomas that appear suddenly without a slow-growing precursor.
Genetic testing pushes the floor lower. For the ~1.3% carrying high-risk inherited mutations, acting on results can eliminate entire categories of risk. A BRCA1 carrier who has preventive breast and ovary removal does not just screen better. She removes the tissue where cancer would develop, cutting breast cancer risk by 90-95% and ovarian cancer risk by ~80%. A Lynch syndrome carrier who starts colonoscopy at 20-25 and has a preventive hysterectomy essentially eliminates uterine cancer risk and catches colorectal precursors decades before they become dangerous. For the remaining ~98.7% without high-risk mutations, polygenic risk scores can refine screening schedules, but the floor does not drop as far.
When genetic testing, prevention, and the full screening stack are combined, residual late-stage cancer risk drops to roughly 1-2.5% over 20 years. As blood testing improves by combining multiple biological signals (DNA, proteins, RNA (the molecules cells use to carry out DNA’s instructions), and metabolic markers (chemicals the body produces that change when cancer is present)) into a single screen, and as AI-assisted imaging improves, the floor will drop further. For now, 1-2.5% with the maximum stack is approximately the best current science can achieve, a dramatic improvement over the 15-20% late-stage risk with no screening at all.
Is it worth ~$3,000/year to lower late stage cancer risk by 10X? For me it is. I also understand that it is a privilege to be able to afford it. I hope that governments will provide this screening to everyone in the future. A world where all cancer is caught early is a world with less fear and heartache, where people can live out their lives to their full potential.





The TALENT trial data is particularly striking. A 2.6% detection rate in never smokers, with 96.5% caught at stage 0 or 1. The fact that family history scaled risk up to 9.1% suggests we should be thinking about lung CT much earlier for high risk families, not waiting for smoking history to qualify.
What services would you recommend genetic testing? I know there is one listed related specifically to brain health but would love to hear an recommendations that cover broader testing.