Transcranial Magnetic Stimulation for Brain Aging, Dementia, and Psychiatric Disorders
What the Science Actually Shows, Where the Field is Headed, and What You Can Do Now
I have been following the Transcranial Magnetic Stimulation (TMS) research closely for several years now, and I wanted to put together a deep dive on where the science actually stands. We also have Dr. Josh Kuluva, a neurologist and psychiatrist specializing in TMS, on NeuroAge’s advisory board and I am thinking about recommendations for TMS for our clients in the future.
As someone who thinks about dementia prevention daily, TMS is one of those technologies that I find genuinely hard to look away from. It is already FDA-cleared for depression, migraines, OCD, smoking cessation, and anxious depression, which means we have real clinical infrastructure and safety data in place. And the emerging research in Alzheimer's disease and cognitive decline, while still early, is some of the most interesting work happening in the neurostimulation space right now. The Sinaptica Phase 2 trial targeting the precuneus in early Alzheimer's patients produced some exciting results. At the same time, I discuss the gaps in what we know. TMS is not yet a proven treatment for dementia, and I have some questions about what repeated magnetic stimulation does to neural circuitry over the long term. Here I walk through all of it, the mechanisms, the clinical evidence, the potential safety concerns, and where I think TMS fits in a broader brain health strategy.
What is TMS?
TMS is a noninvasive brain stimulation technique that uses magnetic fields to gently activate nerve cells in specific areas of the brain, without surgery, drugs, or anesthesia. A figure-8 shaped coil is held against the scalp and sends brief magnetic pulses through the skull. These pulses create tiny electrical currents that stimulate neurons in the brain region directly beneath the coil, and in other connected brain areas. First developed by Anthony Barker in 1985, TMS has grown from a laboratory research tool into a proven medical treatment for several brain and mental health conditions (Koch et al., 2024, Alzheimer’s Drug Discovery Foundation Review).
When TMS pulses are delivered repeatedly in a session, the technique is called repetitive TMS (rTMS). The frequency of the pulses matters. Fast-paced pulses (5 to 20 per second) tend to increase brain activity in the targeted area, while slow pulses (1 per second or fewer) tend to calm it down. A newer approach called theta-burst stimulation (TBS) achieves similar effects in much less time. One version, called intermittent TBS (iTBS), can boost brain activity in roughly three minutes, compared to the 37 minutes needed for a conventional session (van den Heuvel et al., 2024, Biological Psychiatry).
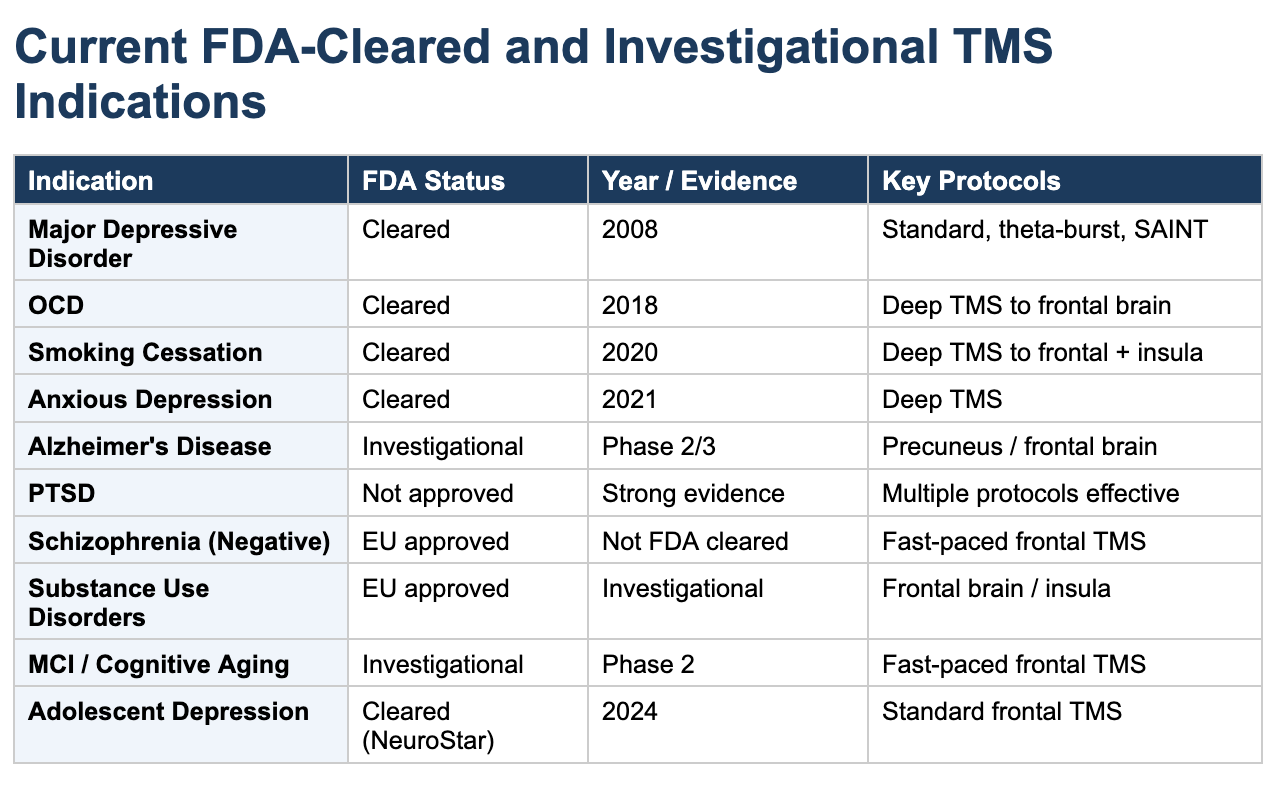
The U.S. Food and Drug Administration (FDA) has cleared TMS for several conditions. Depression received clearance in 2008, followed by migraines, obsessive-compulsive disorder (OCD) in 2018, smoking cessation in 2020, and anxious depression in 2021. Beyond these approved uses, TMS is being actively studied for Alzheimer’s disease, PTSD, schizophrenia, substance use disorders, and age-related cognitive decline (Cotovio et al., 2023, Journal of Clinical Medicine).
How TMS Changes Your Brain, And Is It Safe?
One question I get asked a lot is how a magnetic pulse actually changes the brain. And a fair follow-up, if TMS is strengthening brain connections, could it strengthen connections in areas we don’t want, or even damage neurons through overstimulation?
Here’s what happens at the cellular level. When a TMS pulse reaches the brain, it activates neurons, which triggers a cascade of events. First, it opens receptors on the surface of neurons (called NMDA receptors) that allow calcium to flow in. This calcium influx is the brain’s natural signal to strengthen that connection, the same process that happens when you form a new memory or learn a new skill. This is called long-term potentiation, or LTP, and it’s essentially the molecular basis of learning. TMS induces the expression of AMPA receptor subunits, which in turn trigger the opening of NMDA channels and activate calcium-sensitive cellular pathways that strengthen synaptic connections at both pre- and postsynaptic sites (Antonioni et al. 2025).
Second, TMS triggers the release of BDNF, a protein I think of as plant food for the brain because it helps neurons grow, survive, and form new connections. BDNF is also released during exercise, which one of the ways that exercise can make the brain more healthy. BDNF binds to its receptor (TrkB) and activates pathways that lead to lasting structural changes including new dendritic spines (the tiny branches where neurons communicate), stronger synapses, and increased production of proteins needed for healthy brain function. A 2022 study in Frontiers in Cellular Neuroscience showed that five days of high-frequency TMS enhanced BDNF signaling in the mouse prefrontal cortex and promoted actual physical remodeling of neural connections, including increased spine density and dendritic complexity. The BDNF-TrkB signaling cascade, including the MAPK/ERK and PI3K/Akt pathways, is now understood to be a central mechanism through which TMS produces lasting synaptic changes (Antonioni et al. 2025).
Third, TMS doesn’t just increase brain activity, it also rebalances inhibitory and excitatory signaling. It modulates both glutamate (the brain’s main excitatory chemical) and GABA (the main inhibitory chemical), helping restore a healthy balance. Research in transgenic mouse models has shown that TMS influences GABA receptor function and the synaptosomal-associated protein SNAP25, enhancing GABAergic neuron function (Antonioni et al. 2025). A 2016 study in Nature Communications demonstrated that 10 Hz magnetic stimulation specifically reduces dendritic inhibition onto pyramidal neurons, accompanied by remodeling of postsynaptic gephyrin clusters, providing direct evidence for how TMS recalibrates the excitation-inhibition balance. This is important because many brain disorders involve an imbalance between excitation and inhibition.
Could TMS Cause Unwanted Plasticity or Excitotoxicity?
This is a legitimate scientific question, and it’s one that doesn’t get enough attention in the TMS marketing literature. Excitotoxicity, where too much glutamate overstimulates neurons and kills them, is a real phenomenon that contributes to brain damage in stroke, traumatic brain injury, and neurodegenerative diseases including Alzheimer’s (Wang et al. 2020). So it’s fair to ask whether, if TMS is increasing glutamate signaling and strengthening connections, could it push things too far?
The short answer, based on the current evidence, is that clinical TMS protocols appear to operate well within a safe range, for several reasons. First, the energy delivered by TMS is orders of magnitude below what’s needed to produce the sustained, massive glutamate flood seen in stroke or brain injury. The stimulation is pulsed (not continuous), giving neurons recovery time between pulses. Second, TMS simultaneously activates the brain’s own protective mechanisms. It upregulates GABA (the inhibitory counterbalance), enhances the BDNF pathway (which promotes neuron survival), and increases expression of anti-cell-death proteins. In fact, a 2022 study in Redox Biology found that 25 Hz TMS in an Alzheimer’s mouse model actually reduced glutamate toxicity by increasing production of the glutamate transporter protein GLT-1, which clears excess glutamate from synapses, through activation of the PI3K/Akt pathway. In other words, at least in that model, TMS was anti-excitotoxic rather than pro-excitotoxic. Separately, gene expression analysis has shown that repeated TMS upregulates multiple glutamate and GABA transporter genes, with changes persisting even 10 days after the last stimulation session.
However, dose matters. A 2013 study in hippocampal cell cultures showed that low-intensity magnetic stimulation (1.14 Tesla) promotes healthy dendritic growth, axonal sprouting, and increased synapse density, while high-intensity stimulation (1.55 Tesla) at the same frequency does the opposite, destroying dendrites, causing neuronal lesions, and diminishing synapses. Both effects were mediated through the BDNF-TrkB signaling system. Clinical TMS protocols are carefully calibrated to stay in the growth-promoting range (Chervyakov et al. 2015).
The concern about off-target plasticity, meaning strengthening connections in brain areas you did not intend to stimulate, is theoretically valid but appears to be mitigated by several factors. TMS preferentially activates neurons that are already active or primed to fire, which means it tends to strengthen circuits that are already engaged rather than randomly building new ones. The brain’s own homeostatic mechanisms (essentially, a built-in thermostat that prevents any one circuit from becoming too strong or too weak) also help prevent runaway plasticity. Computational modeling has shown that TMS triggers dose-dependent homeostatic rewiring in recurrent neuronal networks, where inhibitory subpopulations constrain the effects of stimulation and prevent runaway excitation.
That said, most of the long-term safety data comes from animals (mouse brains are quite different than human brains) or relatively short human studies, and the effects of repeated TMS over months or years of maintenance treatment, especially in a brain that is already compromised by disease and may have impaired homeostatic mechanisms, remain genuinely understudied. This is an area that deserves more research attention, and it is one of the main reasons I remain cautious about recommending TMS outside of clinical contexts where it has been FDA approved.
The brain is not a simple circuit board, and the idea that we can magnetically stimulate specific regions without any unintended downstream effects on broader neural wiring is, at best, an assumption that has not been rigorously tested over long time horizons.
TMS as a Window into Brain Aging
As we age, the brain gradually changes. Connections between neurons weaken, the chemical messengers that carry signals between brain cells shift in balance, and the brain becomes less efficient at forming new memories and strengthening neural pathways. These changes show up as the gradual slowing of memory, attention, processing speed, and decision-making that most people experience as they get older. TMS is valuable in this context for two reasons. First, it can measure these age-related brain changes with remarkable precision, and it may also be able to counteract some of them (Freitas et al., 2013, Frontiers in Neuroscience).
Measuring Age-Related Brain Changes
By delivering single or paired magnetic pulses and measuring the brain’s response, researchers can detect how aging affects critical brain functions. Studies show that as we age, the brain becomes less effective at regulating its own activity (a process scientists call cortical inhibition, meaning the brain’s ability to quiet down certain signals so it can focus). The brain also loses some of its ability to communicate efficiently between the left and right hemispheres, and it becomes harder for neurons to strengthen their connections in response to new information, a process known as neuroplasticity. These changes in the brain’s internal “braking” and “communication” systems closely track the cognitive declines people notice in everyday life (Bhandari et al., 2016, Experimental Gerontology).
When TMS is combined with EEG (a brainwave recording technique), researchers can watch in real time how a magnetic pulse travels through the brain’s networks. Think of it like tapping one end of a spiderweb and watching the vibration spread. Recent research shows that these “brain echoes” change noticeably with age, reflecting shifts in both local brain activity and long-distance communication between brain regions. These measurements may be especially good at detecting the very earliest stages of diseases like Alzheimer’s, potentially flagging problems years before any symptoms appear (Battaglia et al., 2021, International Journal of Molecular Sciences).
The Brain Health Index Concept
Alvaro Pascual-Leone’s group at Harvard has proposed the concept of a Brain Health Index (BHI), essentially a “brain fitness score” derived from repeated TMS assessments of how well the brain adapts and rewires over time. The idea is to track each person’s brain plasticity trajectory across their lifespan, much like tracking cholesterol or blood pressure. A declining BHI could flag problems years before any cognitive symptoms emerge, offering a window for early intervention. This concept aligns closely with the growing movement toward preventive brain health, providing an objective measurement that could guide both clinical decisions and research trials (Freitas et al., 2013, Frontiers in Neuroscience).
Enhancing Cognition in Healthy Aging
Several studies have shown that repeated TMS sessions can temporarily sharpen cognitive performance in healthy older adults. Stimulating a region in the front of the brain called the dorsolateral prefrontal cortex (DLPFC), a key area for working memory and decision-making, has been shown to improve working memory. Other protocols have improved the ability to remember to do things in the future (like taking medication at a certain time). These findings suggest that TMS may be able to counteract age-related cognitive slowing by boosting the brain’s ability to strengthen its neural connections, though researchers are still working to determine how long these improvements last and how meaningful they are in daily life (Gomes-Osman et al., 2018, Frontiers in Aging Neuroscience). I would note that the cognitive enhancement results in healthy adults are modest and temporary, and in my view the risk-benefit calculus for using TMS in someone with a healthy brain is very different from using it in someone with a clinical condition.
In animal studies, TMS has been shown to strengthen connections between neurons in the hippocampus (the brain’s memory center) of aging mice. Specifically, TMS boosted levels of BDNF, the plant food for the brain that helps neurons grow, survive, and form new connections. These findings provide a biological explanation for how TMS might preserve brain function in aging, and suggest that the benefits may reach deeper brain structures beyond just the surface area being stimulated (Ma et al., 2014, Neural Regeneration Research).
TMS in Alzheimer’s Disease and Dementia
Alzheimer’s disease (AD) progressively disrupts the brain’s communication networks, particularly a network called the default mode network (DMN). The DMN is active when you recall memories, think about yourself, or let your mind wander. It is one of the first networks to break down in Alzheimer’s. Two of the DMN’s most important hubs, the precuneus and the posterior cingulate cortex, are located deep in the middle of the brain and are among the earliest regions damaged by the amyloid plaques and tau tangles that define Alzheimer’s. TMS offers a way to directly stimulate and support these struggling networks, potentially slowing the disease’s progression.
What the Pooled Research Shows
When researchers combine results from many individual studies (called a meta-analysis), the overall picture for TMS in Alzheimer’s is encouraging. A 2024 meta-analysis by Pagali and colleagues pooled data from 25 randomized controlled trials and found that TMS significantly improved thinking and memory compared to sham (placebo) stimulation. The improvements showed up across all the major cognitive tests used in Alzheimer’s research, including the Mini-Mental State Examination (MMSE), the Montreal Cognitive Assessment (MoCA), and the Alzheimer’s Disease Assessment Scale (ADAS-Cog). The effect sizes were moderate to large, meaning the differences were clinically meaningful, not just statistically significant (Pagali et al., 2024, International Psychogeriatrics).
A 2025 meta-analysis by Zhang and colleagues confirmed these positive findings, while noting that results varied considerably depending on which brain region was targeted, what stimulation settings were used, and what stage of disease the patients were in. The analysis found that targeting the DLPFC (the decision-making and working memory area in the front of the brain) was associated with more consistent positive results, though other targets, including the precuneus and angular gyrus, also showed promise (Zhang et al., 2025, Frontiers in Aging Neuroscience).
A separate 2024 meta-analysis specifically looked at the effects of TMS alone (excluding studies that combined TMS with brain training exercises) in Alzheimer’s patients. Using data from controlled trials published between 2010 and 2023, the analysis found that TMS by itself significantly improved cognitive function. The study also explored which settings worked best, finding that faster-paced pulse protocols (10 to 20 pulses per second) targeting both sides of the DLPFC over 4 or more weeks produced the most consistent cognitive benefits (Li et al., 2024, Journal of Alzheimer’s Disease).
The Sinaptica Phase 2 Trial (The Most Exciting Result So Far)
Perhaps the most significant recent development in TMS for Alzheimer’s comes from a company called Sinaptica Therapeutics. In a year-long study, they tested a personalized form of TMS in patients with mild-to-moderate Alzheimer’s. What made this study special was the precision. Rather than aiming TMS at a generic brain location, Sinaptica used simultaneous TMS and EEG recordings to map each patient’s unique brain activity patterns and fine-tune the treatment to ensure it was actually reaching and activating the default mode network.
The results were striking. After 12 months, patients receiving TMS showed 44% less cognitive decline than patients who received a placebo treatment. The TMS group also maintained their ability to perform daily activities (such as dressing, managing finances, and navigating), showed less cognitive deterioration on standard Alzheimer’s tests, and had more stable mood and behavior, while the placebo group continued to worsen on all measures. Critically, the treatment produced no serious side effects (Koch et al., 2024, CTAD Conference and Alzheimer’s Research & Therapy).
The Sinaptica approach stands out for several reasons. First, instead of targeting the front of the brain like most TMS studies, it targets the precuneus, a deep brain region at the heart of the memory network that breaks down early in Alzheimer’s. Second, it uses real-time brain monitoring (TMS combined with EEG) to verify that the stimulation is actually reaching the right networks and to adjust the dose for each person. Third, the treatment is practical. After an initial two-week start-up phase, patients only need a 20-minute session once a week. Sinaptica’s system received FDA Breakthrough Device Designation in 2022, a label reserved for technologies that may offer significant advantages over existing treatments. The company originally planned to launch a large-scale 280- to 300-person pivotal trial across approximately 20 sites in the U.S. and Europe by the end of 2025. As of early 2026, that pivotal trial had not yet begun enrollment, though Sinaptica completed its Scientific Advisory Board in December 2025 and expanded its partnership with device manufacturer Nexstim, signaling active preparations for trial launch (Sinaptica SAB Announcement, December 2025). In the meantime, Sinaptica initiated a separate Phase 2 study in February 2025 testing a second-generation dual-stimulation system (combining TMS with transcranial electrical stimulation) in up to 60 patients with early-stage Alzheimer’s at multiple centers in Italy. This next-generation approach reduces session times to as little as six minutes (Sinaptica, February 2025, BusinessWire).
These results are encouraging but require important caveats. This was a single Phase 2 trial with a relatively small number of patients. Phase 2 trials are designed to show whether something might work, not to prove that it does. Many interventions that look promising in Phase 2 fail to hold up in larger Phase 3 trials. We also do not yet understand what repeated long-term stimulation of the precuneus does to the broader neural circuitry over years. The brain is not a simple machine where you can turn up activity in one region and expect only the outcomes you intended. Until we have larger, longer, replicated trials, I would characterize these results as hypothesis-generating rather than practice-changing.
Why Target the Precuneus? The Default Mode Network Explained
The shift toward targeting the precuneus reflects a growing understanding that Alzheimer’s may be as much a disorder of broken brain networks as it is of toxic protein buildup. The precuneus acts like a central hub in the brain’s default mode network. Because it connects to so many other regions, stimulating it can send ripple effects throughout the entire memory network. A 2025 trial design from UCLA is exploring this approach using a larger TMS coil to stimulate a broader area of the precuneus. This trial (registered as NCT06597942) is designed to be easier on patients by consolidating sessions, while exploring whether brain imaging and brainwave measurements can confirm that the treatment is engaging the right targets (Leuchter et al., 2025, Alzheimer’s & Dementia TRC&I).
The Moussavi Multisite Trial
The largest randomized controlled trial of TMS in Alzheimer's to date was published in Neurotherapeutics in 2024, led by Zahra Moussavi at the University of Manitoba. The study enrolled 156 patients across hospitals in Canada and Australia, randomizing them into three groups receiving either two weeks of active TMS, four weeks of active TMS, or four weeks of sham TMS, all targeting bilateral DLPFC at 20 Hz. The results were surprising. All three groups, including sham, showed significant cognitive improvement on the ADAS-Cog lasting up to two months, with no significant difference between active and sham treatment at any time point over six months of follow-up. Response rates were nearly identical, around 72 to 73 percent across groups. Rather than concluding that TMS does not work, the authors raised a provocative possibility. Their lab testing confirmed that the Magstim sham coil generates measurable magnetic fields, and they hypothesize it may have been producing a low-level therapeutic current similar to transcranial alternating current stimulation. In other words, there may not have been a true placebo group at all. The study highlights a critical challenge in TMS research, designing sham conditions that genuinely deliver zero stimulation to the brain, and the authors call for future trials using verified inactive sham coils (Moussavi et al., 2024, Neurotherapeutics).
TMS Plus Brain Training, Better Together?
A growing body of evidence suggests that pairing TMS with brain training exercises (like puzzles, memory games, or structured cognitive tasks) produces bigger benefits than either approach alone. A 2024 review of the research found that this combination led to significantly more cognitive improvement than TMS by itself. The reason may be that TMS opens a window of heightened brain plasticity, essentially making the brain more receptive to learning, and the brain training exercises take advantage of that window. The combination may also improve blood flow, neurotransmitter levels, and the brain’s ability to process and metabolize energy (Dong et al., 2024, Frontiers in Aging Neuroscience).
Mild Cognitive Impairment, Catching It Early
TMS has also shown promise for mild cognitive impairment (MCI), the gray zone between normal aging and dementia where people notice memory problems but can still function independently. In a controlled trial, older adults with MCI who received TMS targeting the left DLPFC showed significant improvement in everyday memory tasks (like remembering appointments and where they put things), and these gains persisted a month after treatment ended (Drumond Marra et al., 2015, Behavioural Neurology).
Why Mental Health Is a Pillar of Dementia Prevention
Before diving into TMS for psychiatric conditions, it is worth stepping back to explain why mental health belongs in a discussion about brain aging and dementia at all. The 2024 Lancet Commission on Dementia Prevention, Intervention, and Care identified 14 modifiable risk factors that together account for roughly 45% of dementia cases worldwide. Depression is one of them. The Commission classified depression as a midlife risk factor with a population attributable fraction of about 3%, meaning that if depression were eliminated as a risk factor at the population level, approximately 3% of all dementia cases could theoretically be prevented (Livingston et al., 2024, Lancet Commission on Dementia).
The epidemiological data here are substantial. Meta-analyses consistently find that people with a history of depression have roughly 1.8 to 2 times the risk of developing dementia compared to people without depression. The risk is particularly elevated for vascular dementia, where late-life depression is associated with approximately 2.5 times the risk. A 2025 umbrella review and meta-analysis confirmed that depression in both midlife and late life significantly increases dementia risk, with late-life depression carrying a hazard ratio of 1.95 and midlife depression a hazard ratio of 1.56 (eClinicalMedicine, 2025, Umbrella Review). These are not small effect sizes. For context, these risk elevations are comparable to or larger than many of the other modifiable risk factors for dementia, including physical inactivity and diabetes.
The biological mechanisms connecting depression to neurodegeneration are increasingly well understood. Chronic depression and psychological stress activate the hypothalamic-pituitary-adrenal (HPA) axis, leading to sustained elevations in cortisol. Cortisol is the body’s primary stress hormone and in short bursts is adaptive, sharpening focus and mobilizing energy. But when cortisol stays chronically elevated, it becomes neurotoxic, particularly to the hippocampus, the brain’s memory center and one of the first structures damaged in Alzheimer’s disease. Prolonged cortisol exposure shrinks the hippocampus, reduces BDNF (the same growth factor that TMS boosts), promotes neuroinflammation, accelerates amyloid plaque deposition, and increases tau phosphorylation, all of which are hallmarks of Alzheimer’s pathology (Ouanes and Popp, 2019, Frontiers in Aging Neuroscience).
What makes this especially insidious is the vicious cycle that can develop. Hippocampal atrophy from chronic cortisol exposure impairs the hippocampus’s ability to regulate the HPA axis, which leads to even more cortisol, which causes further hippocampal damage. Longitudinal studies have confirmed that higher plasma cortisol predicts greater hippocampal atrophy over time in people with mild cognitive impairment, and that this atrophy in turn accelerates progression to Alzheimer’s (White et al., 2023, Alzheimer’s and Dementia). A study in cognitively normal midlife adults found that higher serum cortisol was associated with lower total brain volume, reduced glucose metabolism in frontal cortex, and higher amyloid-beta load in Alzheimer’s-vulnerable brain regions (Walhovd et al., 2024, Scientific Reports).
This is not limited to depression. Anxiety disorders, PTSD, chronic psychological stress, and sleep disruption all activate the same HPA axis and cortisol pathways, and all have been associated with increased dementia risk in epidemiological studies. The 2024 Lancet Commission specifically called out treatment-resistant depression as conferring even higher dementia risk than depression that responds to treatment, which raises the question of whether more effective treatment of refractory depression, potentially including TMS, could reduce downstream neurodegeneration.
This is the lens through which I think TMS for psychiatric disorders should be evaluated in the context of brain health. It is not just about symptom relief. Untreated or inadequately treated depression is actively damaging the brain through sustained cortisol exposure, chronic neuroinflammation, reduced BDNF, and accelerated vascular injury. Every month that depression goes unresolved is a month of ongoing neurotoxic insult. If TMS can achieve remission where other treatments have failed, the downstream benefit to long-term brain health could extend well beyond mood improvement. If it were me who was depressed, I would start with exercise (the world’s best antidepressant) and a good talk therapist and then move on to TMS if that wasn’t enough.
TMS in Psychiatric Disorders
Major Depressive Disorder
Depression is the condition where TMS has the longest track record and strongest evidence. International guidelines rate TMS for depression as “definitely effective.” Roughly 40% to 60% of patients with depression achieve full remission with TMS. For people with treatment-resistant depression (meaning their depression has not responded to antidepressant medications), response rates are somewhat lower but still meaningful, especially for people who have run out of other options. A 2024 consensus review endorsed by major depression research centers confirmed that TMS is safe and effective, and that newer, faster protocols may work even better than the original approach (McClintock et al., 2024, Clinical Neurophysiology).
One important question is how long TMS benefits last. A systematic review found that approximately two-thirds of patients remained depression-free at 3 months after treatment, about half at 6 months, and just under half at 12 months. “Maintenance” TMS, where patients return for periodic booster sessions, appears to reduce the risk of relapse, though the optimal schedule for these boosters is still being worked out (Danish Psychiatric Society, 2025, TMS Guidelines).
The Stanford SAINT Protocol (Five Days Instead of Six Weeks)
The Stanford Accelerated Intelligent Neuromodulation Therapy (SAINT) protocol may be the most dramatic innovation in TMS to date. Developed by Nolan Williams and colleagues at Stanford, SAINT compresses what normally takes six weeks of daily treatment into just five days. Patients receive 10 sessions per day, with breaks between sessions, for a total of 90,000 magnetic pulses over the five-day period. What makes SAINT especially sophisticated is that each patient gets a brain scan (functional MRI) beforehand, and the treatment is aimed at the exact spot in their brain’s frontal lobe that is most strongly connected to their mood-regulating circuits (Cole et al., 2020, American Journal of Psychiatry).
In a rigorous clinical trial where neither patients nor doctors knew who was receiving real versus fake treatment, 78% of participants receiving SAINT achieved full remission of their depression, compared to just 13% in the placebo group. In earlier studies, 19 of 21 participants who completed all 50 sessions achieved remission, and 60% were still depression-free a month later. The protocol has also shown the ability to rapidly reduce suicidal thoughts in hospitalized patients, which is especially important given the elevated suicide risk in the weeks following psychiatric hospitalization (Williams, 2022, Psychiatric News).
The FDA cleared the Stanford Neuromodulation Therapy (SNT, the commercial name for SAINT), making it the first FDA-cleared accelerated TMS protocol for depression. Stanford researchers describe the treatment as “retraining” misfiring brain circuits. In many depressed patients, the electrical signals that regulate sadness and emotional control flow in the wrong direction through the brain’s mood circuits, and SAINT appears to correct this (Stanford Medicine, 2025).
How Does TMS Compare to Antidepressants and Therapy?
If you look at the remission numbers for TMS side by side with SSRIs or cognitive behavioral therapy (CBT), they look surprisingly similar at first glance, roughly 30-50% remission across the board. So you might reasonably ask, why bother with TMS.
The answer is that these numbers are not an apples-to-apples comparison. The STAR*D trial, still the largest antidepressant study ever done, found that about a third of patients remitted on the SSRI citalopram as a first-line treatment. If the first drug didn’t work, patients could try up to three more, with each subsequent drug being less likely to help. By the fourth medication, remission rates dropped below 7%. TMS trials, by contrast, are typically enrolling patients who have already failed one or more antidepressants, the harder-to-treat population. The fact that TMS achieves 30-50% remission in treatment-resistant patients, a population where additional medications are barely doing better than placebo, is actually quite impressive. And when you look at meta-analyses of TMS specifically in patients who have failed two or more antidepressants, TMS roughly doubles to triples the odds of remission compared to continuing with medications alone.
Where TMS really shines is the side effect profile. This is not a close contest. SSRIs cause weight gain, sexual dysfunction (in 58-70% of patients), GI problems, insomnia, and withdrawal symptoms on discontinuation. These side effects are significant enough that many patients stop taking their medication. TMS side effects are limited to temporary scalp discomfort and occasional headache at the treatment site. No weight gain. No sexual dysfunction. No GI issues. No withdrawal. No systemic effects at all, because TMS doesn’t enter the bloodstream. It is a targeted, localized treatment. For patients who cannot tolerate medications (pregnant women, the elderly, people with liver or kidney problems), TMS is one of the only effective options available.
This is something I think about a lot. As I’ve written about in my post on optimal cholesterol levels for longevity and brain health, cardiovascular risk factors like high cholesterol and high blood pressure directly damage the brain. High ApoB, high LDL, and elevated blood pressure contribute to white matter injury, the damage to the brain’s wiring that shows up on MRI as white matter hyperintensities and is associated with increased risk of stroke, vascular dementia, and Alzheimer’s. I saw this firsthand in my own brain scans when my total cholesterol was 270 and I was developing white matter hyperintensities (which is part of why I started rosuvastatin despite my primary care doctor telling me I didn’t need it). Many of the systemic side effects of psychiatric medications (weight gain, metabolic changes, elevated blood pressure) can worsen these exact cardiovascular risk factors over time. TMS avoids all of this because it doesn’t enter the bloodstream. This matters especially if you are someone who is already working hard to optimize your metabolic and cardiovascular health and doesn’t want a psychiatric medication undermining those efforts.
That said, TMS is not a magic bullet. It doesn’t work for everyone, it requires multiple clinic visits (unless you’re doing SAINT), and it’s more expensive upfront than a prescription. The emerging evidence suggests that the best outcomes may come from combining TMS with other treatments, whether that is medication, therapy, or lifestyle optimization. TMS plus an SSRI appears to work better than either alone, and TMS combined with psychotherapy has shown remission rates of around 55%, compared to 31-37% for TMS alone.
Obsessive-Compulsive Disorder
In 2018, the FDA cleared deep TMS (dTMS) for OCD treatment, making it the first noninvasive device approved for this condition. OCD involves a loop of brain circuits connecting the cortex (the brain’s outer thinking layer), the striatum (involved in habits and routines), and the thalamus (a relay station deep in the brain). When this loop gets stuck, people experience the repetitive, intrusive thoughts and compulsive behaviors characteristic of OCD. Deep TMS targets this circuit by stimulating areas on top of the brain involved in planning and impulse control. A 2025 systematic review found that four of six controlled trials showed significant reductions in OCD symptoms with real TMS compared to placebo (Aloufi et al., 2025, Cureus).
Multiple reviews of the research have confirmed that TMS works for OCD, though researchers note the need for larger and better-designed studies. TMS is increasingly seen as a valuable add-on treatment for people whose OCD has not responded to standard medications (SSRIs) or therapy. The growing understanding that OCD is a problem of mis-wired brain circuits, rather than a single brain region, has led to more sophisticated treatment approaches that aim to reset the entire dysfunctional loop.
Post-Traumatic Stress Disorder
TMS for PTSD is not yet FDA-approved but has gathered substantial evidence, particularly from military research. A landmark 2025 study of 756 veterans who had both PTSD and depression compared three different TMS protocols head-to-head. All three produced major symptom reductions. Between 63% and 78% of patients responded to treatment, and roughly half achieved full remission, regardless of which protocol they received. This is good news for clinicians because it means they can choose the most practical or comfortable protocol for each patient without sacrificing effectiveness (Philip et al., 2025, Brain Stimulation).
International treatment guidelines currently rate TMS for PTSD as “possibly effective,” a cautious rating that reflects the need for more large-scale controlled studies rather than a lack of positive findings. A recent review confirmed that TMS reduces PTSD severity immediately after treatment, though studies have used different brain targets and protocols, making it harder to draw definitive conclusions about the single best approach. Research is ongoing, with stimulation of the frontal brain regions showing the most promise.
Smoking Cessation and Substance Use Disorders
In August 2020, the FDA cleared a deep TMS device (the BrainsWay H4 coil) to help people quit smoking. In the pivotal trial of 262 participants, 28.4% of those receiving real TMS maintained continuous abstinence for four weeks, compared to just 11.7% in the placebo group. By the end of treatment, 44% had stopped smoking. The treatment targets two brain areas involved in addiction, the prefrontal cortex (involved in decision-making and impulse control) and the insula (a region that processes bodily sensations and cravings) (Dinur-Klein et al., 2014 and BrainsWay FDA Clearance).
Beyond smoking, TMS is being investigated for cocaine, alcohol, methamphetamine, and cannabis use disorders. TMS targeting the decision-making areas of the frontal brain has shown long-lasting reductions in cocaine use, and TMS devices have already received regulatory approval for substance use disorders in Europe. A major review published in Neuropsychopharmacology found that brain stimulation therapies, including TMS, produced meaningful reductions in cravings across multiple types of addiction (Ekhtiari et al., 2023, Neuropsychopharmacology).
Schizophrenia
TMS is being studied for two of schizophrenia’s most challenging symptoms, the “negative” symptoms (apathy, social withdrawal, and difficulty thinking clearly) and auditory hallucinations (hearing voices). A comprehensive review of 48 controlled trials involving over 2,200 patients found that fast-paced TMS targeting the frontal brain may improve negative symptoms, while slow-paced TMS targeting a region near the left ear (involved in language processing) has been studied for reducing hallucinations. While not yet FDA-approved in the U.S., deep TMS has received regulatory approval in Europe for negative symptoms of schizophrenia (Cotovio et al., 2023, Journal of Clinical Medicine).
Adolescent Depression
TMS has also shown promise for teenagers with depression. Recent data show that nearly 60% of adolescents with major depression responded to NeuroStar Advanced TMS treatment. This is especially significant because medication options for teen depression are limited, and antidepressants carry concerns about side effects in younger people (including potential increases in suicidal thinking). The consistent safety record of TMS across studies makes it an appealing option for this age group (Aloufi et al., 2025, Cureus).
How People Can Access TMS Today
TMS for FDA-cleared conditions is widely available across the United States and increasingly around the world. For depression, most major insurance plans now cover TMS after failure of at least one antidepressant medication. Standard treatment involves 30 to 36 sessions, delivered once daily over 6 to 9 weeks. The SAINT/SNT accelerated protocol condenses this into 5 intensive days, though insurance coverage for the accelerated approach varies.
For OCD, treatment involves deep TMS sessions targeting the brain’s habit and compulsion circuits, usually delivered over 4 to 6 weeks. For smoking cessation, the protocol involves 18 sessions over 6 weeks. All TMS treatments are done as outpatient procedures. No anesthesia or sedation is needed, and patients can drive themselves to and from sessions. Side effects are generally mild, most commonly a brief scalp tingling or headache at the treatment site. The risk of seizure is extremely low.
For Alzheimer’s and cognitive decline, TMS is not yet FDA-approved and is generally accessible only through clinical trials. Sinaptica Therapeutics is actively preparing to launch its pivotal Phase 3 trial, and its early-stage Alzheimer’s Phase 2 study is currently enrolling in Italy. Several other academic medical centers also offer TMS for cognitive research, and interested patients or caregivers can search ClinicalTrials.gov for active studies.
TMS for PTSD is used off-label at many VA medical centers. The 2025 multisite VA study of 756 veterans provides strong real-world evidence supporting this practice, and the multiple protocols studied (10 Hz, iTBS, dTMS) all showed comparable effectiveness.
Future Directions and Emerging Applications
Personalized and Precision TMS
The field is rapidly moving toward individualized treatment. Brain scan-guided targeting, as used in both SAINT (for depression) and the Sinaptica approach (for Alzheimer’s), allows clinicians to identify the best stimulation site for each patient’s unique brain, rather than relying on one-size-fits-all placement. Combining TMS with real-time brainwave monitoring (EEG) allows clinicians to see whether the stimulation is actually reaching the intended brain networks and adjust accordingly. Machine learning algorithms are also being developed to predict which patients will respond best to TMS based on their brain scans, genetic profiles, and clinical history.
“Smart” TMS That Adapts in Real Time
Imagine a TMS system that continuously reads your brainwaves during treatment and adjusts the stimulation in real time, like a thermostat that senses the temperature and adjusts the heating. That is the concept behind closed-loop TMS. By monitoring brain activity through EEG and adapting the timing, intensity, and location of pulses moment by moment, these systems could deliver stimulation precisely when and where the brain is most receptive, while avoiding unnecessary stimulation. The Sinaptica system already incorporates some of this approach, and more sophisticated adaptive systems are in development.
Combination Therapies
Combining TMS with medications is an area of intense interest. For Alzheimer’s, Sinaptica CEO Ken Mariash has noted that TMS could someday be paired with the new anti-amyloid drugs (like lecanemab or donanemab) that clear toxic proteins from the brain. For depression, combining TMS with ketamine is being explored, with early evidence suggesting that the two approaches together may enhance brain blood flow and the brain’s ability to rewire itself (Psychiatric Times, 2025). For substance use disorders, combining TMS with behavioral therapies and pharmacotherapy may improve quit rates beyond what either approach achieves alone.
Home-Based and Portable TMS
One of the biggest barriers to TMS is the need for repeated clinic visits, often several times per week for weeks on end. The FDA recently approved ProlivRX, a prescription at-home brain stimulation device for depression that patients can use under physician guidance. As TMS devices become smaller, more affordable, and safer for home use, access could expand dramatically, particularly for “maintenance” booster sessions after an initial in-clinic treatment course. This would be especially beneficial for elderly patients and people with dementia, for whom repeated trips to a clinic are a major burden.
Expanding Neurological Indications
Beyond the conditions discussed above, TMS is being studied for vascular dementia, Lewy body dementia, Parkinson’s disease (both movement and thinking symptoms), stroke recovery, chronic pain, fibromyalgia, multiple sclerosis, and tinnitus (ringing in the ears). Deep TMS already has regulatory approval in Europe for many of these conditions, and ongoing clinical trials continue to build the evidence.
TMS as a Diagnostic Tool
One of the most exciting developments is the growing recognition that TMS is not just a treatment. It is also a powerful diagnostic tool. By measuring how the brain responds to magnetic pulses, researchers can detect characteristic patterns that may distinguish Alzheimer’s patients from healthy individuals, and TMS-derived measures of brain plasticity may serve as early warning signs of cognitive decline risk. Imagine adding a TMS-based “brain plasticity check” to a comprehensive brain health evaluation alongside brain MRI, genetic testing, blood-based biomarkers, and cognitive testing. Together, these tools could give individuals a much clearer picture of their brain health trajectory years before any symptoms appear.
Implications for Brain Health and Dementia Prevention
TMS sits at a fascinating intersection of brain science and clinical medicine. It can both measure brain health and treat brain disorders, making it potentially well-suited to the growing field of brain health optimization and dementia prevention. But I want to close with a dose of realism about where we actually are.
The strongest evidence for TMS is in treatment-resistant depression. For people who have tried medications and therapy and are still suffering, TMS offers a real and well-supported option with a side effect profile that is dramatically better than adding more drugs. The SAINT protocol in particular represents a genuine advance. For depression, I am comfortable saying TMS is a proven treatment, though I would still recommend it after, not instead of, first-line approaches.
For Alzheimer’s and cognitive decline, we are much earlier in the story. The Sinaptica Phase 2 results are the most exciting finding in this space, but Phase 2 is not Phase 3, and the history of Alzheimer’s therapeutics is littered with interventions that looked promising in small trials and failed at scale. We do not fully understand what repeated magnetic stimulation does to neural circuitry over years, and the brain is complex enough that unintended consequences are not just theoretically possible but should be expected until proven otherwise.
The most promising direction may be TMS as a diagnostic and monitoring tool rather than a treatment. Using TMS-EEG to measure brain plasticity over time, as Pascual-Leone’s Brain Health Index concept proposes, carries none of the concerns about unintended neural rewiring and could be genuinely valuable for early detection of cognitive decline risk.
Several other takeaways from the evidence are worth noting. Earlier intervention appears more effective than later. Personalization, whether through brain scan-guided targeting or real-time EEG monitoring, consistently outperforms one-size-fits-all approaches. And combination approaches, pairing TMS with other treatments rather than relying on it alone, tend to produce the best outcomes.
My thoughts in summary
TMS is an area that I’m following with great interest but am not quite ready to pull the trigger on for myself or to recommend to clients for brain aging specifically. I still feel a bit fuzzy on the mechanism of targeted synaptic strengthening and how we can be sure this is happening only in the places in the brain where we’d like it to occur. That said, I wouldn’t be surprised if TMS becomes a standard offering on the time horizon of 2-5 years for brain aging and dementia prevention if it achieves FDA approval for dementia and MCI. It is reasonable to consider TMS for depression and the other indications for which it has been approved now. And for people who have MCI or Alzheimer’s, TMS clinical trials would be something that I would highly recommend because they are likely pretty safe (given FDA approval for other indications) and could offer a solution in a space where solutions are pretty limited.


This is a great overview of TMS, and I appreciate when clinicians explain it as a neuromodulation tool rather than a “brain zap” caricature. From a mechanistic standpoint, what makes TMS interesting is that it’s not trying to “add a chemical,” but to shift network dynamics: patterned magnetic pulses induce small electrical currents in superficial cortex, and repeated sessions can produce longer-lasting changes in excitability and connectivity in circuits implicated in mood and cognition (classically fronto-limbic networks).
A few clinician-facing nuances that are worth highlighting (and that patients often ask about):
1. Where it fits best: strongest evidence is for major depressive disorder (especially treatment-resistant), and it can be a valuable option when meds are poorly tolerated or insufficient.
2. Time course: it’s not instantaneous for most; improvements often accrue over weeks, and the trajectory matters as much as the endpoint.
3. Safety profile: generally favorable; the commonest issues are scalp discomfort/headache early on. Seizure risk is low but real, so screening and protocol adherence matter.
4. Precision matters: coil placement, intensity, frequency (e.g., rTMS vs theta-burst), and individualized targeting can meaningfully influence response; this isn’t one-size-fits-all.
I also like when posts name the “real-world” tradeoff: TMS can be time-intensive, but for the right patient the benefit can be life-changing, especially when paired with psychotherapy and sleep/circadian stabilization to consolidate gains.
Thanks for making a high-signal, low-hype explainer!